The FDA has approved Moderna's RSV vaccine for older adults.
Politeness: Modern
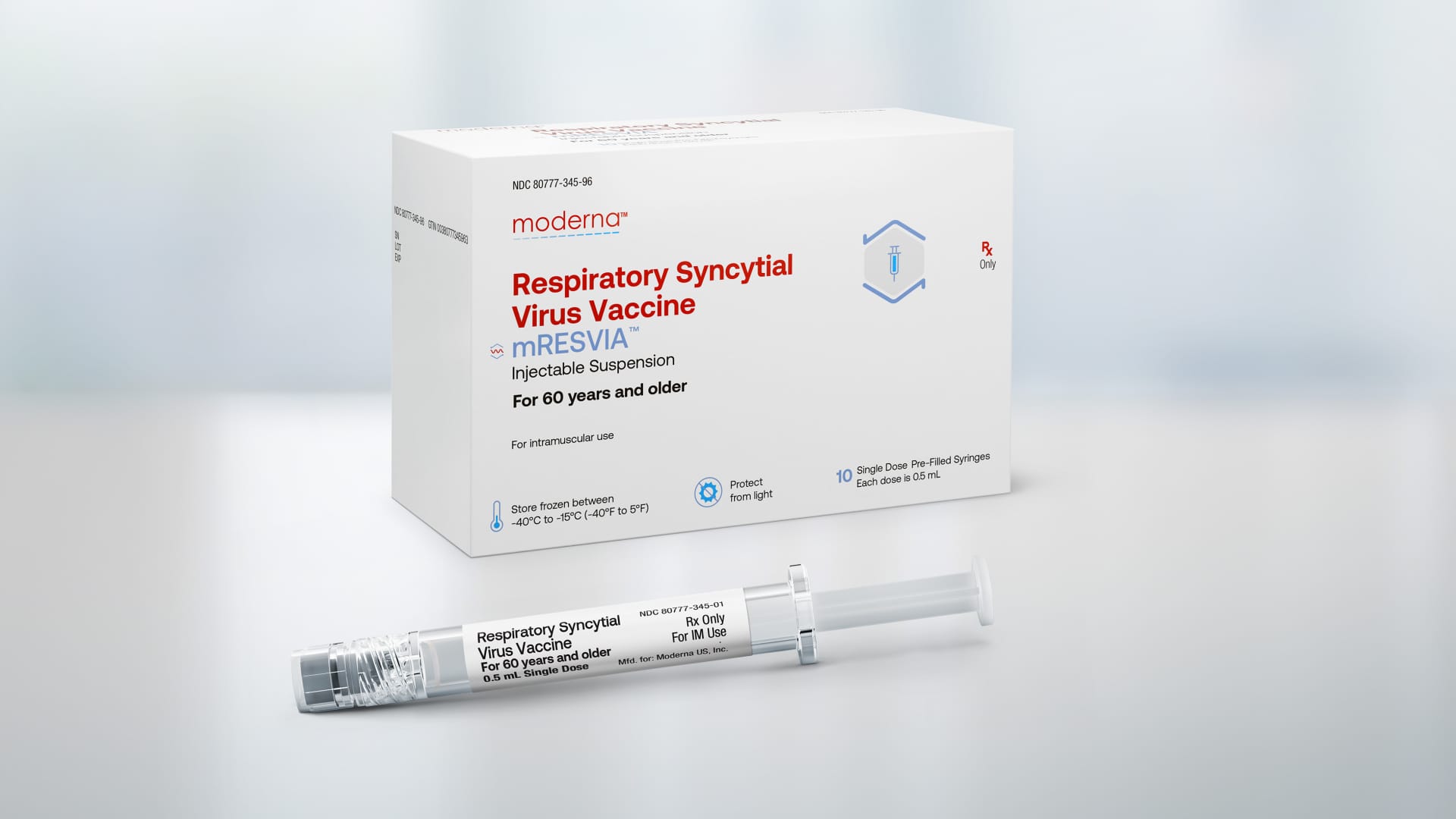
The Food and Drug Administration approved on Friday Modern Respiratory syncytial virus vaccine for adults ages 60 and older, the company's second product ever to hit the U.S. market.
The decision is a victory for Moderna, which urgently needs another source of revenue amid falling demand for its Covid vaccine, its only commercially available product.
The approval of Moderna's vaccine is based on an advanced-stage study of older adults, who are more vulnerable to severe cases of RSV. The virus kills between 6,000 and 10,000 seniors each year and leads to 60,000 to 160,000 hospitalizations, according to data from the Centers for Disease Control and Prevention.
Moderna's vaccine is marketed under the brand name mRESVIA. It is the first messenger RNA vaccine approved for a disease other than Covid. The company's vaccine is also the only RSV vaccine available in a prefilled syringe, making it easier to administer to patients.
A CDC advisory panel will vote in June on recommendations for the use and targeting of the Moderna vaccine. The company expects a recommendation equivalent to that for existing RSV vaccines from GSK And PfizerModerna executives said during a conference call on May 1.
A positive recommendation from the CDC would allow Moderna's vaccine to compete against GSK and Pfizer, which launched their respective vaccines in the U.S. last fall. Pfizer's vaccine has lagged GSK's so far, but both vaccines have generated hundreds of millions in sales so far.
Moderna's full-year 2024 revenue forecast of around $4 billion includes revenue from its RSV vaccine.
The approval shows how versatile Moderna's messenger RNA platform is beyond treating Covid. The biotech company is using this technology to fight a number of different diseases, including RSV, cancer and a highly contagious gastrointestinal virus called norovirus.
“The FDA approval of our second product, mRESVIA, builds on the strength and versatility of our mRNA platform,” Moderna CEO Stéphane Bancel said in a press release. “With mRESVIA, we continue to make a difference for patients by addressing global public health threats related to infectious diseases.”
The biotech company currently has more than 40 products in development, several of which are in advanced testing phases. This includes its combination vaccine against Covid and flu, which could be approved as early as 2025.
More health reports from CNBC
Moderna is also developing, among other things, a standalone flu vaccine, a personalized cancer vaccine together with Merck, and vaccines against latent viruses.
Moderna says it expects sales growth again in 2025 and expects to break even in 2026 thanks to the introduction of new products.
Investors have high hopes for the long-term potential of Moderna's mRNA product pipeline: The company's shares have risen more than 60% this year after falling nearly 45% in 2023.
Data from vaccine studies
The FDA was originally scheduled to make a decision on Moderna's vaccine on May 12, but the agency delayed approval, citing internal “administrative constraints.”
A Phase 3 trial of about 37,000 people showed that Moderna's vaccine was 83.7 percent effective at preventing at least two symptoms of RSV after about three months. New data from that trial in February showed that the vaccine's effectiveness dropped to 63 percent after 8.6 months.
At the time, these results raised concerns among investors that the vaccine's effectiveness would wane faster than vaccines from GSK and Pfizer. Moderna said in a statement that comparisons were not possible without head-to-head studies of the vaccines.
The company added that its study had different study populations, geographic locations and case definitions for RSV, among other things.
No significant safety concerns were identified among patients who received the shot as part of the study. Most side effects were mild to moderate and included pain at the injection site, fatigue, headache, muscle aches and joint pain.