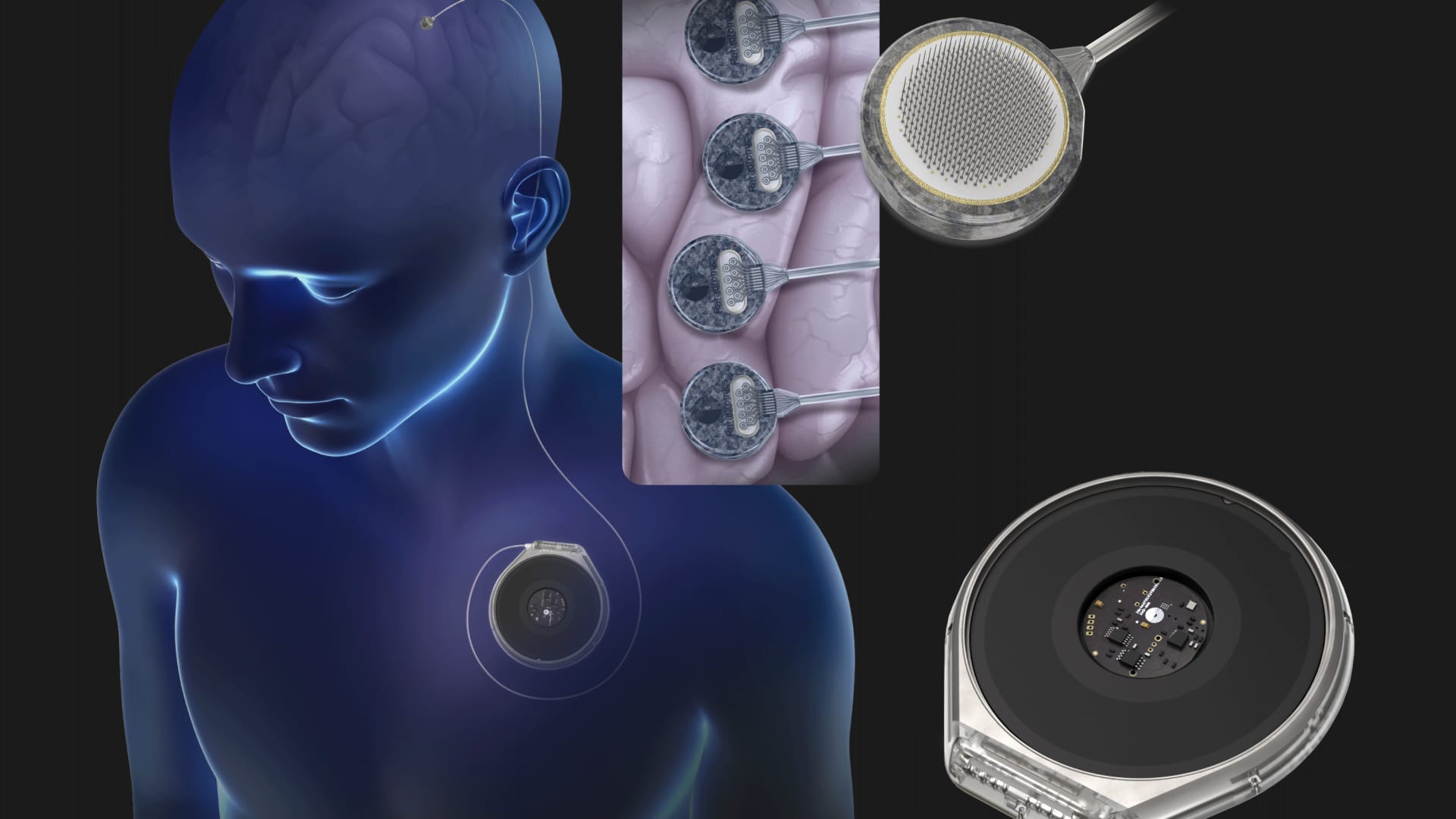
Neurotech startup Paradromics will test its brain implant next year as the race for the top spot in brain-computer interfaces heats up.
“The brain is a super fascinating organ. We have about 85 billion neurons and each neuron is a million times slower than a computer chip. And yet the brain does incredible things,” said Matt Angle, CEO and founder of Paradromics, in an interview with “CNBC Tech: The Edge.”
“This means that if you want to get data in and out of the brain, you need to be able to communicate with a lot of different neurons at the same time. And that's where the focus comes from on building these high-speed, high-data-rate devices,” he added.
The trial followed rival Neuralink's attempt to implant a chip into a patient's brain in March. The company, co-founded by Elon Musk, later announced that part of its brain implant malfunctioned in the weeks following the procedure.
Founded in 2015, Paradromics has raised $87 million in venture capital and $18 million in public funding to date. The Austin, Texas-based startup expects the devices to retail for about $100,000 a unit.
“Paradromics' mission is to transform otherwise untreatable brain diseases into solvable technology problems. Essentially, we are building a medical device that addresses unmet needs,” Angle said.
While Angle expects the device to treat a wide range of conditions, Paradromics will initially focus on patients who have lost their ability to communicate, whether due to paralysis, a disease such as amyotrophic lateral sclerosis (also known as ALS) or a spinal cord injury.
“We decided to focus on motor and language because these topics are widely discussed in our research community and the scientific evidence on them is available,” said Vikash Gilja, Paradromics’ chief scientific officer.
“Paradromy can leverage science and apply the right engineering to take us from research to medical device,” he added.
Gilja told CNBC that the device is powered wirelessly and does not need to be charged.
“The only thing you have to do as a user is to go through a short calibration routine to learn the mapping of electrical signals to intentions. But once that mapping is learned, the system can be used,” Gilja said.
Angle hopes that Paradromics will receive commercial approval to sell the product as early as 2029.
“We expect that the first million people who have brain-computer interfaces will use them to treat serious diseases,” Angle said.
“I think in 20 years the discussion might be different and some of these devices might be suitable for consumers as well. In the meantime, however, we are really focused on building safe, reliable and robust devices for people with physical and mental disabilities.”