Drugmakers are betting that direct radiation to tumors will be the next big breakthrough in cancer.
Bristol Myers Squibb, AstraZeneca, Eli Lilly and other pharmaceutical companies have spent about $10 billion on deals to acquire or partner with radiopharmaceutical manufacturers. They have snapped up smaller startups to get their hands on a technology that, although still in its infancy, could treat numerous types of cancer.
“Any large company that has a business in oncology, or for which oncology is an important therapeutic category, probably needs to get involved in this area in one way or another,” said Michael Schmidt, an analyst at Guggenheim Securities.
Two radiopharmaceuticals from Novartis are already available. Another few dozen are in development, according to Schmidt's count. It's difficult to estimate the overall market opportunity because there are so many possible cancers that the drugs could treat, he said.
Schmidt believes the category could reach sales of under $5 billion if the technology remains limited to treating some cancers, such as prostate cancer and neuroendocrine tumors If it turns out to be in the tens of billions effective on more types of cancer.
The drugs work by attaching radioactive material to a target molecule that looks for and binds to a specific marker on cancer cells. The trick is finding markers that exist on cancer cells but not on healthy cells. This can allow the treatment to deliver radiation to cancer cells and relieve the rest of the body from radiation exposure Damage associated with many cancer drugs.
It took time to prove that the technology could work both scientifically and financially. The first radiopharmaceuticals were approved in the early 2000s. But interest from big pharmaceutical companies only recently increased.
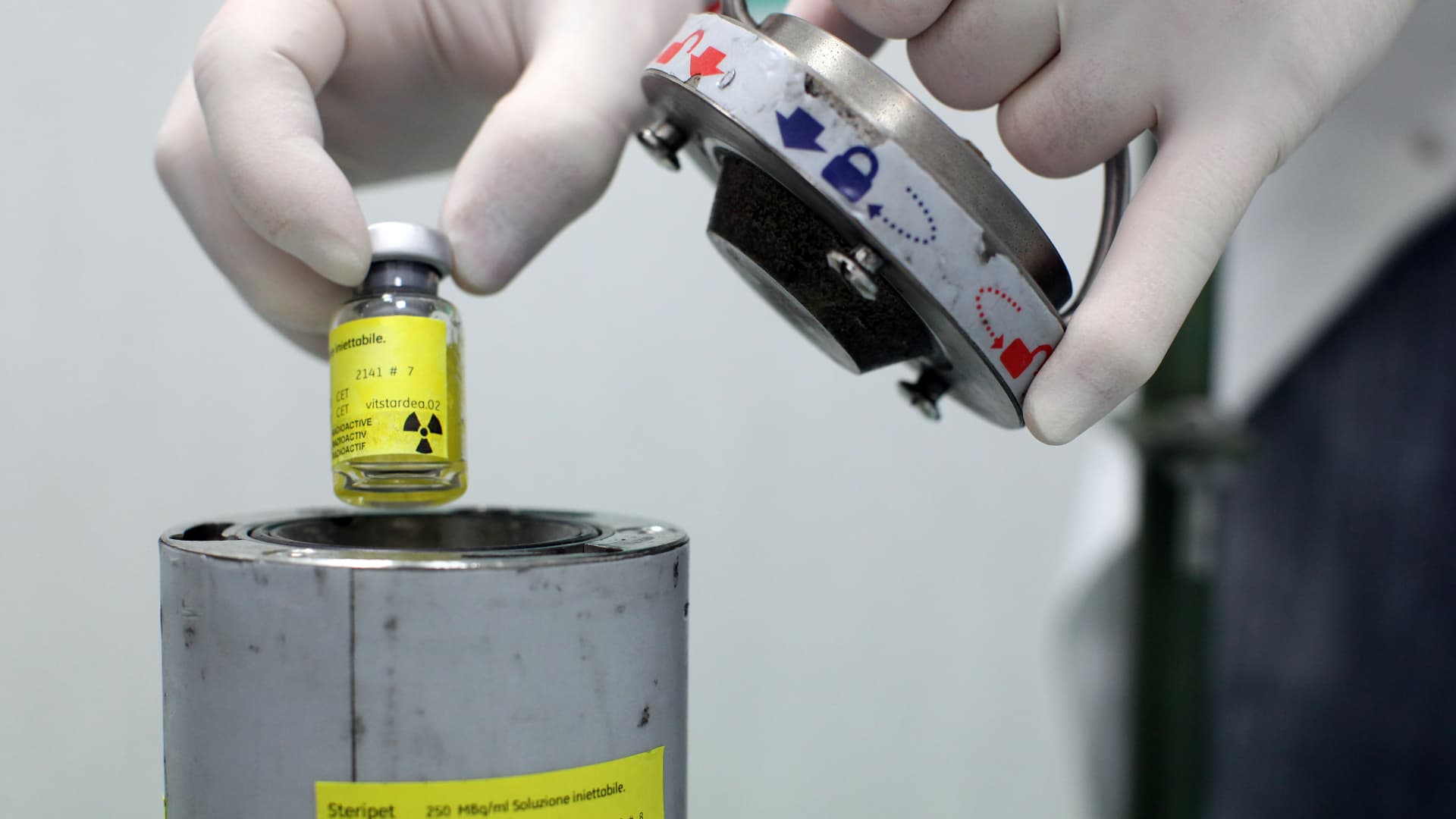
An employee works at the NSA radiopharmaceutical plant in Aedea, Rome, Italy.
Franco Origlia | Getty Images
Producing the drugs requires complex manufacturing and logistics, two major drawbacks. Radioactive material decays quickly, so patients need to be treated within a few days of treatment.
Pharmaceutical companies have proven they can manage complex, time-sensitive drugs like CAR-T for blood cancers or gene therapies for rare diseases. Then Novartis showed that these strategies could be applied to radiopharmaceuticals.
The Swiss pharmaceutical giant received approval in 2018 for a radiopharmaceutical drug called Lutathera against a rare type of cancer in the pancreas and gastrointestinal tract. In 2022, Novartis secured further approval for the drug Pluvicto against prostate cancer. The drugs combined are expected to reach about $4 billion in sales by 2027, according to FactSet consensus estimates.
These successes sparked broader interest in radiopharmaceuticals.
“We put all of this together and thought we should do something, we need to do business here,” said Jacob Van Naarden, president of Eli Lilly's oncology business.
Lilly acquired radiopharmaceutical maker Point Biopharma for about $1.4 billion last year and also signed some partnerships with companies developing the treatments. One of the most important factors in Lilly's initial search was whether the companies were willing to make the drugs, Van Naarden said. Radiopharmaceuticals are not easy to manufacture, and Lilly wanted to ensure that any initial acquisition could produce the drugs itself rather than outsourcing the work.
Manufacturing was also a key component in Bristol Myers Squibb's $4.1 billion acquisition of RayzeBio, said Ben Hickey, president of RayzeBio. At the time of the acquisition, RayzeBio was nearing completion of a factory in Indiana and had secured its own supply of radioactive material needed to develop the experimental drugs in its pipeline.
“It was clearly one of the criteria to make sure we were in control of our own destiny,” Hickey said.
Novartis has shown why this is so important, as the company initially struggled to produce enough Pluvicto doses. The company is investing more than $300 million to open and expand radiopharmaceutical manufacturing facilities in the United States so that the drug can be manufactured and quickly made available to patients. The company is now able to meet demand for the treatment, which requires careful distribution planning.
Victor Bulto, president of Novartis' U.S. business, said each dose is equipped with a GPS tracker to ensure it gets to the right patient at the right time. Novartis is transporting doses to destinations within nine hours of the factory to minimize the risk of disruption from storms, Bulto said.
Doctors and patients on the receiving end also feel the complexity.
The Bassett Healthcare Network in New York state had to improve its medical certification to handle radioactive material before administering Lutathera and Pluvicto, Dr. Timothy Korytko, chief of radiation oncology at Bassett. The intravenous medications must be administered by a certified specialist.
It may take several weeks from the time a radiopharmaceutical is prescribed until it is administered. For Pluvicto, patients come once every six weeks for up to six treatments.
Radiopharmaceuticals begin to break down as soon as they are manufactured, so they only have a shelf life of a few days.
Ronald Coy and his wife Sharon.
Courtesy: Ronald Coy
Ronald Coy knows how important it is to show up for his appointments. Coy, a retired firefighter who has been battling prostate cancer since 2015, drives more than an hour across New York state to receive Pluvicto in Bassett. Coy hasn't had any problems so far, but he's worried a snowstorm could derail one of his dates through the end of January.
“Hopefully we don’t get any major storms by then, or if we do, it’ll be a week before I travel,” Coy said.
When Coy returns home from treatment, he must take precautions such as staying away from his wife Sharon so that she is not exposed to radiation. He drinks a lot of water to remove extra radiation from his body. He doesn't mind a few days of minor inconvenience when it comes to fighting his cancer.
For Novartis, investments in the infrastructure for the production and distribution of radiopharmaceuticals would be worthwhile for Pluvicto and Lutathera alone, said Bulto. But it's even more attractive because it has the potential to treat more types of cancer. As an example, he cites Novartis' work to develop a drug for a marker This occurs in 28 different tumors, including breast, lung and pancreatic cancer.
“If we were able to take all of these insights that we've developed from a manufacturing distributor and put them into the service of patients with lung cancer and patients with breast cancer and potentially demonstrate these levels of meaningful efficacy and tolerability, then we're talking about one very big potential impact on cancer treatment and of course also a very profitable business,” he said.
More CNBC Health coverage
At this point, it's still an if. The field is still in its infancy, executives say, and the potential of radiopharmaceuticals beyond the current cancers they treat has yet to be proven.
“If we can expand the target and tumor type repertoire, this could be a very large class of drugs,” Eli Lilly’s Van Naarden said, adding that it was hard to say at this point whether the class would be “super important.” will be. or “just important”.
Bristol Myers Squibb sees an opportunity in combining radiopharmaceuticals with existing cancer drugs such as immunotherapy, said Robert Plenge, Bristol's chief research officer. AstraZeneca shares this vision.
AstraZeneca spent $2 billion to acquire Fusion Pharmaceuticals earlier this year. Susan Galbraith, the company's executive vice president of oncology research and development, points to existing therapies that combine immunotherapy with radiation.
How large AstraZeneca's radiopharmaceutical portfolio ultimately becomes will depend on its initial prostate cancer program and other undisclosed targets already in the works, Galbraith said. However, she believes the technology will become an important part of cancer drugs in the next decade.
It could take years to understand the true potential of the technology, as many experimental drugs are still in the early stages of development. An open question is whether other radiopharmaceuticals are as safe and well tolerated as Novartis' Pluvicto, particularly those that use other types of radioactive material, Guggenheim analyst Schmidt said.
Ronald Coy has been battling prostate cancer for almost 10 years. He started taking Novartis' Pluvicto earlier this year.
Courtesy: Ronald Coy
Big pharmaceutical companies aren't waiting to jump into the race. Stories like Coy's encourage them that the work will be worth it.
Over the course of nearly a decade, Coy was treated multiple times for prostate cancer that had spread to his bones. After just one treatment of Pluvicto earlier this year, blood tests showed that Coy's cancer rates had plummeted.
Not everyone responds so well to Pluvicto, and things could always change for Coy. But for now, Coy is happy to be part of the group that is responding well to Pluvicto. It's worth the drive and the precautions for him.
“I feel very lucky every day to be part of – as it stands now – the Third, where it’s working really well for me,” he said.
—CNBC's Leanne Miller contributed to this report.